|
1/14/2024 0 Comments Mn element charge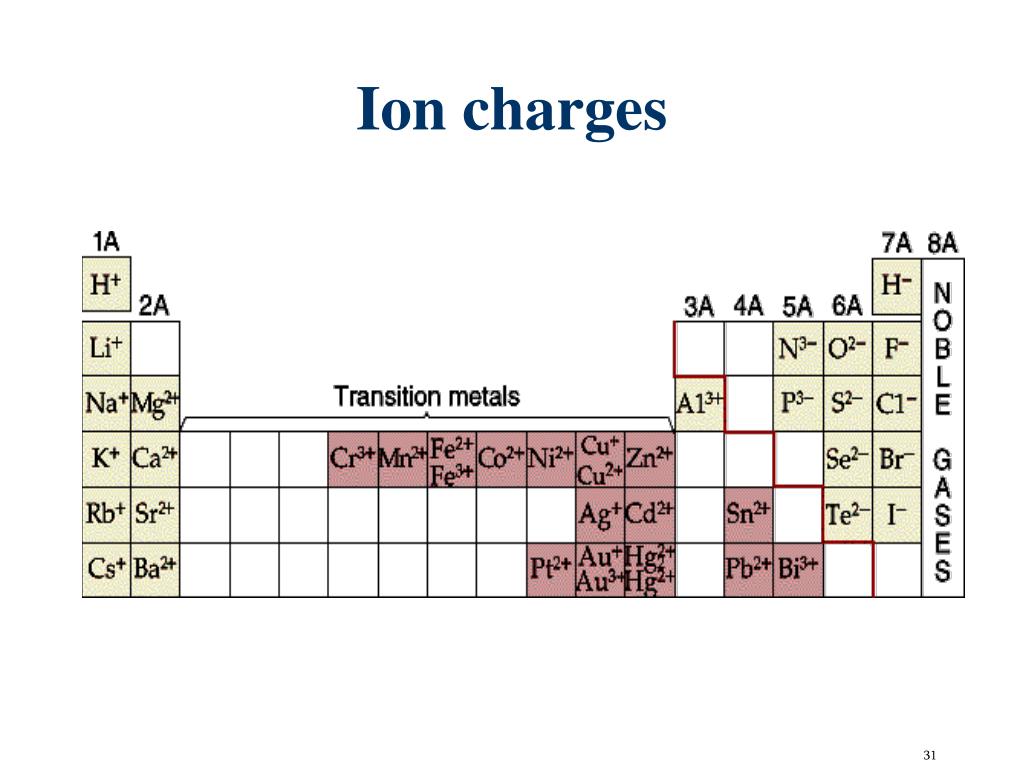
Note: Greek prefixes are not used to indicate the number of atoms of each element in the formula unit for the compound (e.g., FeI 3 is named "iron(III) iodide" not "iron(III) triiodide"). 
The anion is named by adding the suffix -ide to the root of the (nonmetal) element name (e.g., iodine = I, " iodide" = I - sulfur = S, " sulfide" = S 2-). The charge on the cation is indicated using a Roman numeral, within parentheses, immediately following the name of the cation (e.g., Fe 3+ = "iron(III)", Fe 2+ = "iron(II)"). Manganese (Mn) is a transition element having various charges, and an atomic number of 25. And again, you know that the ionic charge of O is 2. In Mn 2 O 3, the Manganese (Mn) is bonded to Oxygen (O). 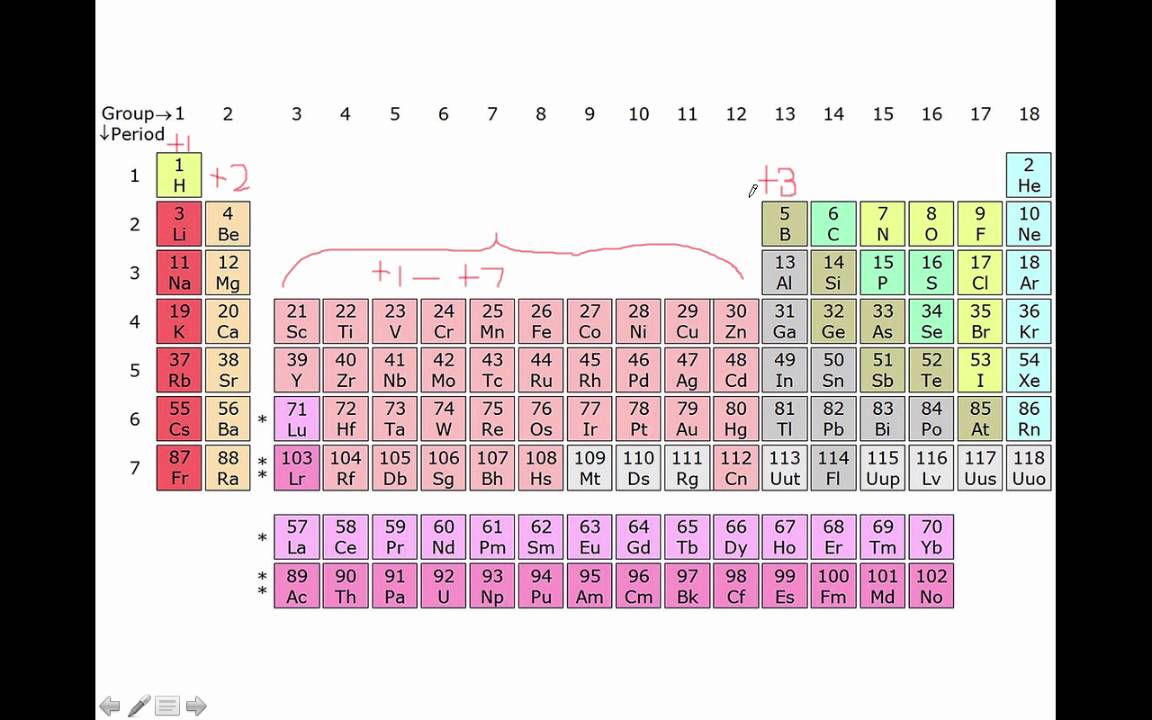
A number of forms of manganese occur in nature ( 300 minerals). Fixed Charge The charge is always the same value based on electron configuration. Manganese is the 12th most abundant element and 3rd most abundant transition metal (cf. Hence the ionic charge of Manganese in MnCl 2 is 2+. Definitions: ionic compound: metal + nonmetal(s) (eg. So you can easily say that the ionic charge of Mn should be 2+, then only it will get canceled out. The name of the cation is the same as the name of the (neutral metal) element from which it is derived. For an atom in elemental form (an element standing alone with no charge). You know that the ionic charge of Cl is 1. The positive ion (cation) is written first in the name the negative ion (anion) is written second in the name. For example, iron(III) iodide, FeI 3, is composed of iron ions, Fe 3+ (elemental iron is a metal), and iodide ions, I - (elemental iodine is a nonmetal). A binary ionic compound is composed of ions of two different elements - one of which is a metal, and the other a nonmetal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
